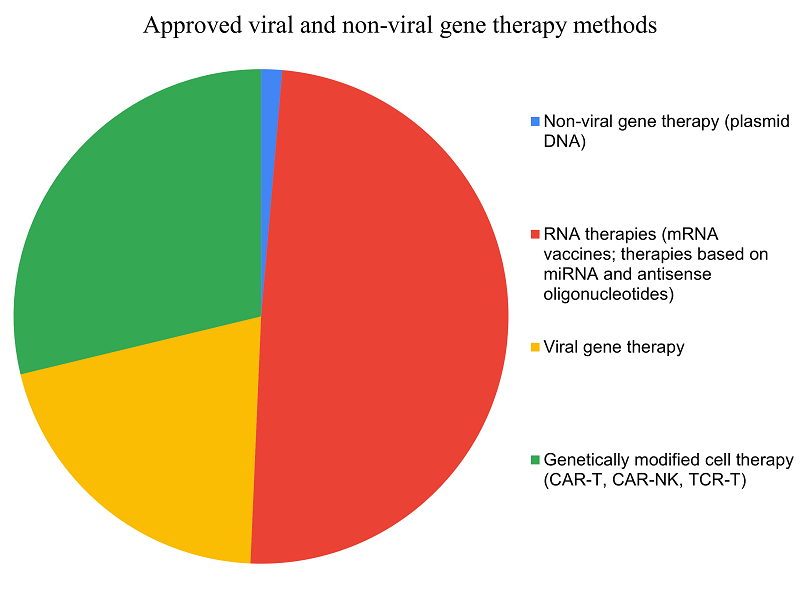

Figure 1. Approved drugs using viral and non-viral gene therapy methods as of the second quarter of 2025 (distribution shown in %) [1] where: 1.4% – non-viral gene therapy (pDNA); 49.3% – RNA drugs (mRNA vaccines; drugs based on siRNA and antisense oligonucleotides); 20.5% – viral gene therapy; 28.8% – gene-modified cell therapy (CAR-T, CAR-NK, TCR-T).