Identification of Androgen Deficiency in Infertility and Reduced Ovarian Reserve Based on HPLC-MS/MS and IHLA Measurements
Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, 4 bld. 2 Oparina str., Moscow, 117513 Russia, *e-mail: s_kindysheva@oparina4.ru.
Keywords:HPLC-MS; IHLA; androgen deficiency states
DOI:10.18097/BMCRM00182
The androgen deficiency and associated states represent is an important problem that affects the quality of women live. The most widely the androgen influence has been studied in the reproductive period in relation to polycystic ovary syndrome; however about laboratory methods to determine the impact of androgen deficiency and its clinical manifestation in the case of young women with a reduced ovarian reserve and with infertility are still actively discussed. Clinical medicine still needs generally approved markers of androgen deficiency states and its lower reference values. In this work we illustrate the perspective of measurements of steroid hormones panel to verify the diagnosis on the basis of high performance liquid chromatography with tandem mass spectrometry and immunochemical methods.
|
CLOSE

|
Table 1.
The results of the steroid profile MS measurement for patients with androgen deficiency(AD) and control group.
|
|
CLOSE

|
Table 2.
The results of the steroid profile IHLA measurements for patients with androgen deficiency and control group.
|
|
CLOSE

|
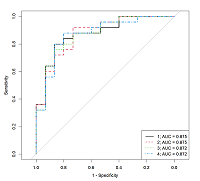
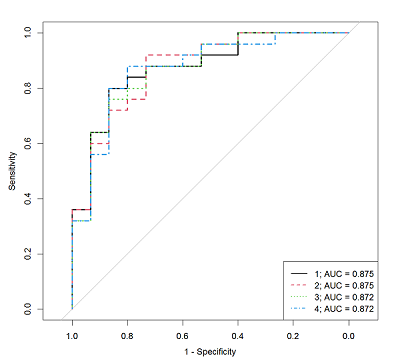
Table 3.
The characteristics of the logistic regression models that allow us to determine the androgen deficiency presence by the steroid hormones concentrations measured by IHLA in the patient serum.
|
|
CLOSE

|
Table 4.
The parameters of the logistic regression models that allow us to determine the androgen deficiency presence by the steroid hormones concentrations measured by IHLA in the patient serum.
|
|
CLOSE

|
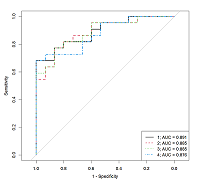
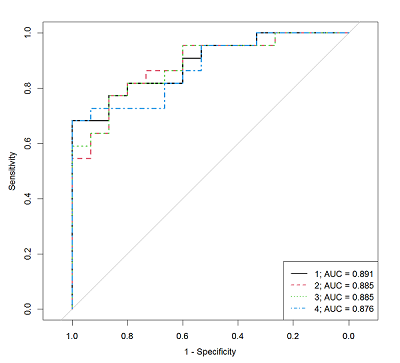
Table 5.
The characteristics of the logistic regression models that allow us to determine the AD presence by the steroid hormones concentrations measured by HPLC-MS/MS method in the patient serum.
|
|
CLOSE

|
Table 6.
The parameters of the logistic regression models that allow us to determine the androgen deficiency presence by the steroid hormones concentrations measured by HPLC-MS/MS in the patient serum.
|
FUNDING
This work was performed within state assignment «Solving the problem of infertility in modern conditions with the aid of a clinical diagnostic model of infertile marriage development and innovate technologies in assisted reproduction programs» No.121040600410-7.
Supplementary materials are available at http://dx.doi.org/10.18097/BMCRM00182
REFERENCES
- Neves, A.R., Montoya-Botero, P., Polyzos N.P. (2022) Androgens and diminished ovarian reserve: the long road from basic science to clinical implementation. A comprehensive and systematic review with meta-analysis. American Journal of Obstetrics and Gynecology, 227(3), p.401-413.e18. DOI
- James, A. (2008) Androgens and Reproductive Aging. Fertility and Sterility, 89(1), 261. DOI
- Bonne, C., Saurat, J. H., Chivot, M., Lehuchet, D., Raynaud, J.P. (1977) Androgen receptor in human skin. The British journal of dermatology, 97(5), 501-503. DOI
- Davis, S.R., Davison, S.L., Donath, S., Bell, R.J. (2005) Circulating androgen levels and self-reported sexual function in women. JAMA, 294(1), 91-96. DOI
- Walters, K.A. (2015) Role of androgens in normal and pathological ovarian function. Reproduction, 149(4), R193-R218. DOI
- Burger, H.B. (2002) Androgen production and mechanisms. Androgen production in women. Fertility and Sterility, 77(4), 3-5. DOI
- Mathur, R., Glenn D.B. (2010) Androgen deficiency and therapy in women. Current Opinion in Endocrinology Diabetes and Obesity, 17(4), 342-349. DOI
- Chernukha, G.E., Naidukova, A.A., Udovichenko, M.A., Kaprina, E.K., Ivanets, T.Yu. (2019) Androgen profile in patients with polycystic ovary syndrome and its association with metabolic dysfunction. Obstetrics and gynecology, 11, 122–128. DOI
- Tabeyeva, G.I., Shamilova, N.N., Zhakhur, N.A., Pozdnyakova, A.A., Marchenko, L.A. (2013) Premature ovarian failure is an enigma of the 21st century. Obstetrics and gynecology, 12, 16–21. DOI
- Karashima, Sh., Osaka, I. (2022) Rapidity and Precision of Steroid Hormone Measurement. Journal of Clinical Medicine, 11(4), 956. DOI
- Handelsman, D.J. (2017) Mass spectrometry, immunoassay and valid steroid measurements in reproductive medicine and science, 32(6), 1147-1150. DOI
- Kanakis, G.A., Tsametis, Ch.P, Goulis, D.G. (2019) Measuring testosterone in women and men. Maturitas. 125, 41-44. DOI
- Trabert B., Xu, X., Falk R.T., Guillemette, Ch., Stanczyk, F.Z. and McGlynn, K.A. (2015) Assay reproducibility of serum androgen measurements using liquid chromatography-tandem mass spectrometry. J. Steroid Biochen. Mol. Biol., 155, 56-62. DOI
- Gavisova, A.A., Dolgushina, N.V., Frankevich, V.E., Ivanets, T.Yu., Gus, A.I., Nazarenko, T.A., Sukhikh, G.T. (2022) FAD – female androgen deficiency, Russian State Patent Agency Certificate, No. 2022124063 of 13.09.2022. DOI
- Davis, S.R. (2002) Androgen deficiency states and sequellae. When to suspect androgen degiciency other than at menopause.Fertility and Sterility. 77, 68-71. DOI
- Traish, A.M., Morgentaler, A. (2022) Androgen Therapy in Women with Testosterone Insufficiency: Looking Back and Looking Ahead. Androgen: Clinical Research and Therapeutics. 3(1), 1-13. DOI
- Jeanneret, F., Tonoli, D., Rossier, M.F., Saugy, M., Boccard, J. (2016) Evaluation of steroidomics by liquid chromatography hyphenated to mass spectrometry as a powerful analytical strategy for measuring human steroid perturbations. J. Chromatogr. A., 1430, 97–112. DOI
- Andrew, R., Homer N.Z.M. (2020) Mass spectrometry: Future opportunities for profiling and imaging steroids and steroid metabolites. Curr. Opin. Endocr. Metab. Res., 15, 71–78. DOI
- Ferchaud, V., Courcoux, P., Bizec, B.L., Monteau, F., Andre, F. (2000) Enzymatic hydrolysis of conjugated steroid metabolites: Search for optimum conditions using response surface methodology. Analyst. 125(12), 2255–2259. DOI
- Gomes, R.L., Meredith, W., Snape, C.E., Sephton, M.A. (2009) Analysis of conjugated steroid androgens: Deconjugation, derivatisation and associated issues. J. Pharm. Biomed. Anal. 49(5), 1133–1140. DOI
- Fang, K., Pan, X.-J., Huang, B., Liu, J.-L., Wang, Yu, Gao, J.-P. (2010) Progress on Keto Groups Derivatization of Steroid Hormones in Gas Chromatography-Mass Spectrometry Analysis. Chinese J. Anal. Chem. 38(5), 743–751. DOI
- Luque-Córdoba, D., López-Bascón, M.A., Priego-Capote, F. (2020) Development of a quantitative method for determination of steroids in human plasma by gas chromatography–negative chemical ionization–tandem mass spectrometry. Talanta, 220, 121415, DOI
- Son, H.H., Yun, W.S., Cho, S.-H. (2020) Development and validation of an LC-MS/MS method for profiling 39 urinary steroids (estrogens, androgens, corticoids, and progestins). Biomed. Chromatogr. 34(2), 1–16. DOI
- Tudela, E., Deventer, K., Geldof, L., Eenoo, P.V. (2015) Urinary detection of conjugated and unconjugated anabolic steroids by dilute-and-shoot liquid chromatography-high resolution mass spectrometry. Drug Test. Anal. 7(2), 95–108. DOI
- Deventer, K., Pozo, O.J., Verstraete, A.G., Eenoo, P.V. (2014) Dilute-and-shoot-liquid chromatography-mass spectrometry for urine analysis in doping control and analytical toxicology. TrAC - Trends Anal. Chem., 55, 1–13. DOI
- Dittadi, R., Matteucci, M., Meneghetti, E., Ndreu, R. (2018) Reassessment of the Access Testosterone chemiluminescence assay and comparison with LC-MS method. J. Clin. Lab. Anal. 32(3), 1–5. DOI
- Kanakis, G.A., Tsametis, Ch.P., Goulis, D.G. (2019) Measuring testosterone in women and men. Maturitas., 125, 41-44. DOI
- Taylor, A., Keevil, B., Huhtaniemi, I.T. (2015) Mass spectrometry and immunoassay; how to measure steroid hormones today and tomorrow. European Journal of Endocrinology, 173, D1–D12. DOI
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Retrieved 2018, from: https://www.R-project.org/