The 40th Anniversary of the Institute of Physiologically Active Compounds of the Russian Academy of Sciences
1Institute of Physiologically Active Compounds of the Russian Academy of Sciences, 1 Severny proezd, Moscow region, Chernogolovka, 142432 Russia;*e-mail: pushkarev@ipac.ac.ru
2Department of Physics, M.V. Lomonosov Moscow State University, 1 Leninskie Gory, Moscow, 119991 Russia
3P.N. Lebedev Physical Institute, Russian Academy of Sciences, 53 Leninsky prosp., Moscow, 119333 Russia
Key words: phthalocyanines; rare earth elements; erbium; sandwich complexes; luminescence; near IR region/p>
DOI: 10.18097/BMCRM00029
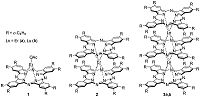
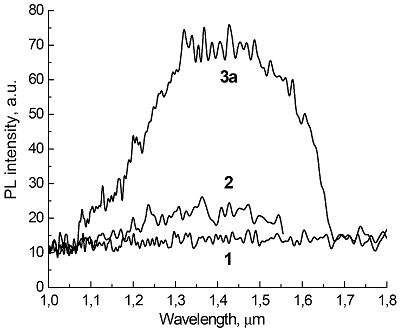
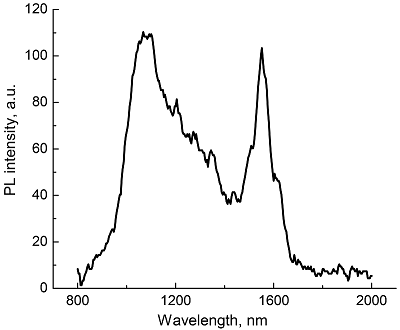
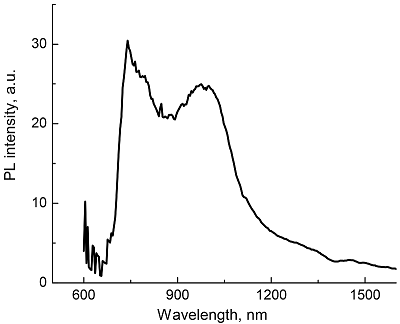
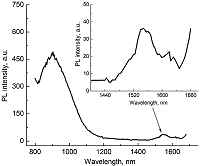
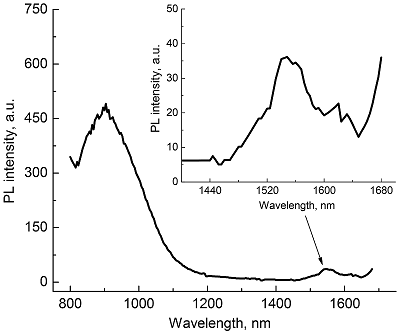
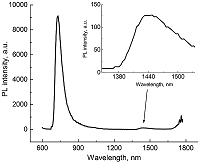
Composite materials based on polystyrene and ErIII phthalocyaninates of single-, double- and triple-decker structure were obtained, and their spectral luminescent characteristics in the near-IR (NIR) range were studied. All the ErIII complexes in these composites reveal 4f photoluminescence (PL), which is observed at 1550 nm for mono- and tris(phthalocyaninate), whereas in the case of bis(phthalocyaninate), the PL maximum is shifted to the short-wavelength region and appears at 1440 nm. A comparative analysis of the properties of composites and individual phthalocyanine compounds in films and solutions was carried out. In the case of a single-decker complex, this emission was detected for the first time in the polystyrene matrix, while for individual ErIII mono(phthalocyaninates), this process was not observed earlier.
ACKNOWLEDGEMENTS
This work was supported by RFBR and the Government of Moscow (Grant No. 15-32-70017) and the Council under the President of the Russian Federation for State Support of Young Scientists and Leading Scientific Schools (Grant MD-2991.2017.3) and performed within the framework of the State Assignment of 2018 (Theme 45.5 Creation of compounds with given physicochemical properties).
REFERENCES
- Kao, K. C., Hwang, W. (1981). Electrical Transport in Solids, With Particular Reference to Organic Semiconductors, Pergamon.
- Myers, J. D., Xue, J. (2012). Organic Semiconductors and their Applications in Photovoltaic Devices. Polymer Reviews, 52(1), 1-37. DOI
- Lattante, S. (2014). Electron and hole transport layers: their use in inverted bulk heterojunction polymer solar cells. Electronics, 3, 132-164. DOI
- Hiramoto, M., Kubo, M., Shinmura, Y., Ishiyama, N., Kaji, T., Sakai, K., Ohno T., Izaki, M. (2014). Bandgap science for organic solar cells. Electronics, 3, 351-380. DOI
- Leznoff, C. C., Lever, A.B.P. Editors (1989-1996). Phthalocyanines – properties and applications, VCH, New York.
- Kadish, K.M., Smith, K.M., Guilard, R. Editors (2010-2012). Handbook of porphyrin science: with applications to chemistry, physics, materials science, engineering, biology and medicine, World Scienti?c, Singapore.
- Jiang, J. Editor (2010). Functional phthalocyanine molecular materials. In Structure and Bonding, 135, Springer.
- Belogorokhov, I.A. (2014). Semiconductor materials of the 21st century from phthalocyanines and nanosilicon, LAP LAMBERT Academic Publishing.
- Pushkarev, V.E., Nemykin, V.N., Tomilova, L.G. (2016). Historic overview and new developments in synthetic methods for preparation of the rare-earth tetrapyrrolic complexes. Coordination Chemistry Reviews, 319, 110–179. DOI
- Belogorokhov, I.A., Ryabchikov, Yu.V., Tikhonov, E.V., Pushkarev, V.E., Breusova, M.O., Tomilova, L.G., Khokhlov, D.R. (2008). Photoluminescence in semiconductor structures based on butyl-substituted erbium phthalocyanine complexes. Semiconductors, 42(3), 321–324. DOI
- Pakhomov, G.L., Kuzin, E.E., Murel, A.V. (2006). NIR photoresponse in the mixed phthalocyanine films. Central European Journal of Physics, 4(4), 494–502. DOI
- Belogorokhov, I.A., Mamichev, D.A., Pushkarev, V.E., Tomilova, L.G., Khokhlov, D.R. (2010). Luminescent properties of semiconductor composite systems composed of erbium triphthalocyanine molecules and a silicon slot structure in the near-infrared region. JETP Letters, 92(10), 676–680. DOI
- Smola, S.S., Snurnikova, O.V., Fadeyev, E.N., Sinelshchikova, A.A., Gorbunova, Yu.G., Lapkina, L.A., Tsivadze, A.Yu., Rusakova, N.V. (2012). The first example of near-infrared 4f luminescence of sandwich-type lanthanide phthalocyaninates. Macroheterocycles, 5(4-5), 343–349. DOI
- Wang, R., Zhang, F. (2014). NIR luminescent nanomaterials for biomedical imaging. Journal of Materials Chemistry B, 2(17), 2422–2443. DOI
- Pushkarev, V.E., Breusova, M.O., Shulishov, E.V., Tomilov, Yu.V. (2005). Selective synthesis and spectroscopic properties of alkyl-substituted lanthanide(III) mono-, di-, and triphthalocyanines. Russian Chemical Bulletin, International Edition, 54(9), 2087–2093. DOI
- Berkovits, V.L., Ziminov, A.V., Kazanskii, A.G., Kolos’ko, A.G., Ramsh, S.M., Terukov, E.I., Fenukhin, A.V., Ulin, V.P., Yurre, T.A., Kleider, J.P. (2007). Influence of the molecular structure of copper phthalocyanines on their ordering in thin films and photoluminescence and absorption spectra. Physics of the Solid State, 49(2), 272–277. DOI
- Ishikawa, N., Sugita, M., Tanaka, N., Ishikawa, T., Koshihara, S., Kaizu, Y. (2004). Upward temperature shift of the intrinsic phase lag of the magnetization of bis(phthalocyaninato)terbium by ligand oxidation creating an s = 1/2 spin. Inorganic Chemistry, 43(18), 5498–5500. DOI
- Ishikawa, N., Okubo, T., Kaizu, Y. (1999). Spectroscopic and quantum chemical studies of excited states of one- and two-electron oxidation products of a lutetium triple-decker phthalocyanine complex. Inorganic Chemistry, 38(13), 3173–3181. DOI