Comparative Analysis of the Proteomic Profile of HaCaT Keratinocytes Using a 1DE Concentrating Gel
Institute of Biomedical Chemistry, 10 Pogodinskaya str., Moscow, 119121 Russia; e-mail: juliaks@bk.ru
Keywords:keratinocytes НаСаТ; SDS extracts; 1DE-gel concentration, in-gel tryptic digestion, SearchGUI; X! Tandem; MS-GF+, proteins encoded by genes of chromosome 18; LC-MS/MS
DOI:10.18097/BMCRM00180
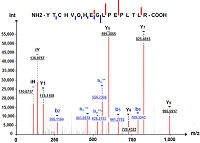
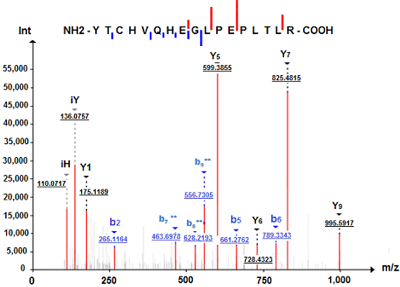
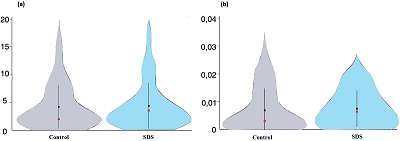
A sample preparation protocol for proteomic profiling of HaCaT keratinocyte was evaluated. It is based on protein solubilization in the presence of 0.2% SDS, a 1DE-gel concentration procedure (SDS-PAGE without fractionation in the separating gel), and in gel cleavage by trypsin followed by LC-MS/MS. Using tandem mass spectrometry with electrospray ionization, a comparative analysis of HaCaT keratinocyte proteins was carried out before and after exposure of cells to sodium dodecyl sulfate (25 mg/ml) for 48 hours; proteins encoded by human chromosome 18 genes were chosen as the comparison proteins. A total of 2418 proteins were detected in the НaСaT immortalized human keratinocytes, 70% of these proteins were identified by two or more unique peptides. Panoramic mass spectrometry analysis identified 38 proteins encoded by chromosome 18 genes, 27 proteins were common to control НaСaT cells and НaСaT cells exposed to SDS. Using the Metascape database (https://metascape.org), an enrichment analysis of GO terms of the Biological Process category of chromosome 18 gene encoded proteins of НaСaT keratinocytes was performed before and after the SDS exposure. The SDS exposure resulted in a slight enrichment of the GO term "response to stimulus" (GO:0050896) and the related GO term "negative regulation of biological process" (GO:0048519). We found decreased expression levels of membrane proteins encoded by chromosome 18 genes related to cell-cell adhesion (GO:0098609), such as DSC1, DSC3, and DSG1. A decrease in the expression level of desmosomal cadherins is characteristic of malignant neoplasms developing from epithelial tissue cells of various internal organs, mucous membranes, and skin. The method of preparation of НaСaT keratinocyte samples used in this work increased the sensitivity of proteomic analysis of cell culture and made it possible to identify twice as many proteins in one gel strip as compared to the number of proteins (1284) in НaСaT samples subjected to osmotic shock and cleavage by trypsin in solution.
|
CLOSE

|
Table 1.
The list of proteins encoded by human chromosome 18 genes and identified in control and SDS-treated samples of HaCaT human keratinocytes.
|
FUNDING
The work was done in the framework of the Russian Federation fundamental research program for the long-term period for 2021-203 (№122030100168-2). Mass-spectrometry measurements were performed using the equipment of “Human Proteome” Core Facilities of the Institute of Biomedical Chemistry (Russia).
Supplementary materials are available at http://dx.doi.org/10.18097/BMCRM00180
REFERENCES
- Ramadan, Q., Ting, F.C. (2016) In vitro micro-physiological immune-competent model of the human skin. Lab. Chip, 16(10), 1899-1908. DOI
- OECD (2013), Test No. 431: In Vitro Skin Corrosion: Reconstructed Human Epidermis (RHE) Test Method, OECD Publishing, Paris,34 p. DOI
- OECD (2013), Test No. 439: In Vitro Skin Irritation - Reconstructed Human Epidermis Test Method, OECD Publishing, Paris,21 p. DOI
- Rusanov, A.L., Luzgina, N.G., Lisitsa, A.V. (2017) Sodium Dodecyl Sulfate Cytotoxicity towards HaCaT Keratinocytes: Comparative Analysis of Methods for Evaluation of Cell Viability. Bulletin of Experimental Biology and Medicine, 163(2), 284-288. DOI
- Lindberg, M., Forslind, B., Sagstrom, S., Roomans, G.M. (1992) Elemental changes in guinea pig epidermis at repeated exposure to sodium lauryl sulfate. Acta Dermato-Venereologic, 72(6), 428–431. DOI
- Miura, Y., Hisaki, H., Fukushima, B., Nagai,T., Ikeda, T. (1989) Detergent induced changes in serum lipid composition in rats. Lipids, 24(11), 915–918. DOI
- Van de Sandt, J.J., Bos, T.A., Rutten, A.A. (1995) Epidermal cell proliferation and terminal differentiation in skin organ culture after topical exposure to sodium dodecyl sulphate. In Vitro Cell. & Dev. Biol. Animal, 31(10), 761–766. DOI
- Petushkova, N.A., Rusanov, A.L. , Zgoda, V.G., Pyatnitskiy, M.A., Larina, O.V., Nakhod, K.V., Luzgina, N.G., Lisitsa, A.V. (2017) Proteome of the human hacat keratinocytes: identification of the oxidative stress proteins after sodium dodecyl sulpfate exposure. Molecular Biology, 51(5), 748–758. DOI
- Quirino, J.P. (2018) Sodium dodecyl sulfate removal during electrospray ionization using cyclodextrins as simple sample solution additive for improved mass spectrometric detection of peptides. Anal Chim Acta, 16 (1005), 54-60. DOI
- Shevchenko, A., Tomas, H., Havlis, J., Olsen, J.V., Mann, M. (2006) In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc., 1(6), 2856-60. DOI
- Gold Biotechnology (2018) In-gel digestion and extraction of proteins protocol. Retrieved September 9, 2022 from: https://goldbio.com/documents/1060/In%20Gel%20Digestion%20and%20Extraction%20of%20Proteins%20Protocol.pdf
- Kachuk, C., Stephen, K., Doucette, A. (2015) Comparison of sodium dodecyl sulfate depletion techniques for proteome analysis by mass spectrometry. J. Chromatography A. DOI
- Ilavenil, S., Al-Dhabi, N.A., Srigopalram, S., Kim, Y.O., Agastian, P., Baaru, R., Choi, K.C., Arasu, M.V., Park, C.G., Park, K.H. (2016) Removal of SDS from biological protein digests for proteomic analysis by mass spectrometry. Proteome Sci., 14, 11. DOI
- Shkrigunov, T., Pogodin, P., Zgoda, V., Larina, O., Kisrieva, Y., Klimenko, M., Latyshkevich, O., Klimenko, P., Lisitsa, A., Petushkova, N. (2022) Protocol for increasing the sensitivity of MS-based protein detection in human chorionic villi. Curr. Issues Mol. Biol., 44 (5), 2069–2088. DOI
- UniProt: the Universal Protein Knowledgebase in 2023. The UniProt Consortium. Nucleic Acids Research, 51 (D1), D523–D531. DOI
- Kisrieva, Y.S., Samenkova, N.F., Larina, O.B., Zgoda, V.G., Karuzina, I.I., Rusanov, A.L., Luzgina, N.G.,Petushkova, N.A. (2020) Comparative study of the human keratinocytes proteome of the HaCaT line: identification of proteins encoded by genes of 18 chromosomes under the influence of detergents. Biomeditsinskaya Khimiya, 66(6), 469-476. DOI
- Walker, J.M. (1994) The bicinchoninic acid (BCA) assay for protein quantitation. Methods Mol. Biol., 32, 5–8. DOI
- Chambers, M., Maclean, B., Burke, R. et al. (2012) A cross-platform toolkit for mass spectrometry and proteomics. Nat. Biotechnol., 30, 918–920. DOI
- Vaudel, M., Barsnes, H., Berven, F.S., Sickmann, A, Martens, L. (2011) SearchGUI: An open-source graphical user interface for simultaneous OMSSA and X!Tandem searches. Proteomics, 11(5), 996–999. DOI
- Vaudel, M., Burkhart, J., Zahedi, R. et al. (2015) PeptideShaker enables reanalysis of MS-derived proteomics data sets. Nat Biotechnol., 33, 22–24. DOI
- Florens, L., Carozza, M. J., Swanson, S.K., Fournier, M., Coleman, M.K., Workman, J. L., Washburn, M.P. (2006) Analyzing chromatin remodeling complexes using shotgun proteomics and normalized spectral abundance factors. Methods., 40(4), 303-311. DOI
- Ashburner, M., Ball,C.A., Blake, J.A., Botstein, D., Butler, H., Cherry, J.M., Davis, A.P., Dolinski, K., Dwight, S.S., Eppig, J.T., Harris, M.A., Hill, D.P., Issel-Tarver, L., Kasarskis, A., Lewis, S. & Matese, J.C., Richardson, J.E., Ringwald, M., Rubin, G.M., Sherlock, G. (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat. Genet., 25(1), 25-9. DOI
- Mi, H., Thomas, P. (2009) PANTHER pathway: an ontology-based pathway database coupled with data analysis tools. Methods Mol Biol., 563, 123-40. DOI
- Kopylov, A.T., Zgoda, V.G., Archakov, A.I. (2009) Label-free quantitative analysis of proteins using mass-spectrometry. Biomeditsinskaya Khimiya, 55(2), 125-39. DOI
- Uhlén, M., Fagerberg, L., Hallström, B.M., Lindskog C. , Oksvold, P., Adil Mardinoglu, A., Sivertsson, Å,. Kampf, C., Sjöstedt, E.(2015). Tissue-based map of the human proteome. Science, 347 (6220). DOI
- Oldach, M. (2018) Normalized spectral abundance factor (NSAF) for quantitative liquid chromatography mass spectrometry-based proteomics. GitHub. Retrieved September 9, 2022 from: https://github.com/moldach/proteomics-spectralCount-normalization ' target='_blank' > DOI
- Eisenberg, E., Levanon, EY. (2013) Human metang genes, revisited. Trends Genet., 29, 569–574. DOI
- Hounkpe, B.W., Chenou, F., de Lima, F., De Paula, E.V. (2021) HRT Atlas v1.0 database: redefining human and mouse housekeeping genes and candidate reference transcripts by mining massive RNA-seq datasets. Nucleic Acids Res., 49 (D1), 947-955. DOI
- Lane, L., Argoud-Puy, G., Britan, A., Cusin, I., Duek, P.D., Evalet, O., Gateau, A., Gaudet, P., Gleizes, A., Masselot, A., Zwahlen, C., Bairoch, A. (2012) neXtProt: a knowledge platform for human proteins. Nucleic Acids Res., 40 (Database issue), D76-83. DOI
- Wu, Q., Feng, Q., Xiong, Y., Xing, L. (2020) RAB31 is targeted by miR-26b and serves a role in the promotion of osteosarcoma. Oncol. Lett., 20(5), 244. DOI
- Tanaka, Ki, Kanazawa, I., Richards, J.B., Goltzman, D, Sugimoto, T. (2020) Modulators of Fam210a and Roles of Fam210a in the Function of Myoblasts. Calcified Tissue International, 106, 533–540 DOI
- Poulton, C.J., Schot, R., Kia, S.K., Jones, M., Verheijen, F.W., Venselaar, H., Marie-Claire, de Wit Y., de Graaff, E., Bertoli-Avella, A.M., Mancini, G.M.S. (2011) Microcephaly with simplified gyration, epilepsy, and infantile diabetes linked to inappropriate apoptosis of neural progenitors. Am. J. Hum. Genet., 89(2), 265-76. DOI
- Hall, P.A., Russell, S.E.H. (2004) The pathobiology of the septin gene family. J. Pathol., 204(4), 489-505. DOI
- Dolat, L., Hunyara, J.L., Bowen, J.R., Spiliotis, E.T. (2014) Septins promote stress fiber-mediated maturation of focal adhesions and renal epithelial motility. Journal of Cell Biology, 207(2), 225-35. DOI
- Montagna, C., Bejerano-Sagie, M., Zechmeister, J.R. (2015) Mammalian septins in health and disease. Res. Rep. Biochem., 5, 59–73. DOI 10.2147/RRBC.S59060
- Farrugia, A.J., Rodrıguez, J., Orgaz, J.L., Lucas, M., Sanz-Moreno, V. & Calvo, F. (2020) CDC42EP5/BORG3 modulates SEPT9 to promote actomyosin function, migration, and invasion. J. Cell Biol., 219 (9), e201912159. DOI
- Zhou,Y., Zhou B., Pache, L., Chang, M., Khodabakhshi, A.H., Tanaseichuk, O., Benner, C., Sumit, K., Chanda, S.K. (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nature Communication, 10(1), 1523. DOI
- Heath, C.G., Viphakone, N., Wilson, S.A. (2016) The role of TREX in gene expression and disease. Biochem J., 473(19), 2911–35. DOI
- Dominguez-Sanchez, M.S., Saez, C., Japon, M.A., Aguilera, A., Luna, R. (2011) Differential expression of THOC1 and ALY mRNP biogenesis/export factors in human cancers. BMC Cancer, 11(77). DOI
- Huber, O., Petersen, I. (2015) 150th anniversary series: desmosomes and the hallmarks of cancer. Cell Commun Adhes., 22(1), 15–28. DOI
- Takeda, A., Kajiya, A., Iwasawa, A., Nakamura, Y., Hibino, T. (2002) Aberrant expression of serpin squamous cell carcinoma antigen 2 in human tumor tissues and cell lines: evidence of protection from tumor necrosis factor mediated apoptosis. Biol. Chem., 383, 1231–1236. DOI
- Tonnetti, L., Netzel-Arnett, S., Darnell, G.A., Hayes, T., Buzza, M.S., Anglin, I.E., Suhrbier, A., Antalis, T.M. (2008) SerpinB2 protection of retinoblastoma protein from calpain enhances tumor cell survival. Cancer Res., 68, 5648–5657. DOI
- Ding, S., Blue, R.E., Morgan, D.R., Lund, P.K. (2014) Comparison of multiple enzyme activatable near-infrared fluorescent molecular probes for detection and quantification of inflammation in murine colitis models. Inflamm. Bowel Dis., 20(2), 363-77. DOI
- Askew, Y.S., Pak, S.C., Luke, C.J., Askew, D.J., Cataltepe, S., Mills, D.R., Kato, H., Lehoczky, J., Dewar, K., Birren, B., Silverman, G.A. (2001) SERPINB12 is a novel member of the human ov-serpin family that is widely expressed and inhibits trypsin-like serine proteinases. J. Biol. Chem., 276(52), 49320-30. DOI